 
We leave it out inĪgain, if you want to emphasize that H + is hydrated, then you can write: In solution as well, with no role in the actual reaction. Since AgF is a solid, weĪre saying that it precipitates from the reaction, and it wouldn't be Note that HF is a weak acid, so we leave it together. Separating the aqueous strong electrolytes, we have: Note that all strong bases contain a metal,Īnd all salts contain either a metal or ammonium. This is a matter of memorizing the seven strong acids and checking for the presence ofĪ metal or ammonium (NH 4 +). NaCl, KBr, MgCl 2, and many, many more, all containing metals or NH 4. HCl, HBr, HI, HNO 3, HClO 3, HClO 4, and H 2SO 4 Delete any ions that appear on both sides of the equation.Ĭlearly rule 2 is the tricky one. 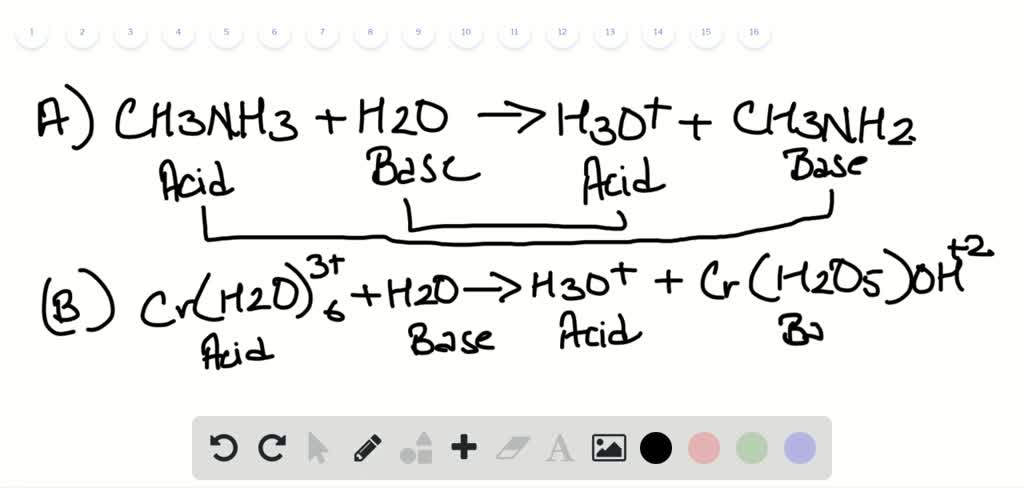
Only consider breaking up the (aq) substances.The three rules for writing net ionic equations are really quite straightforward. First of all, we MUST start with an equation that includes the physical state: Writing net ionic equtaions is easier than you might think. If you want to emphasize that H + is hydrated, then you can write: Where we have neglected the Na + and Cl because theyĪre not really involved. They areįloating around at the beginning and still floating around at the end. Notice that Na + and Cl never really react. (For more information on classifying electrolytes, click here.) As such, they dissociateĬompletely into their ions in solution, and although we might write "HCl" we HCl, NaOH, and NaCl are all strong electrolytes. Spectator ions that really don't get involved.įor example, consider the reaction described by the following full molecular equation: That focus on the principal substances and ions involved in a reaction-the principal species-ignoring those One of the most useful applications of the concept of principal species While you may already print out the reaction summary (assuming you are using registered version), we will look at the printout when discussing stoichiometry calculations.The reason to write a chemical equation is to express what we believe isĪctually happening in a chemical reaction. You may export the equation as TXT, as RTF (rich text format) for use in any wordprocessor, as HTML to put it on the website, as UBBC (universal bulletin board code) to post it on some forum or as LaTeX to use it in a LaTeX prepared paper. Balancing chemical equations was never as easy before!īalancing chemical redox equations and net ionic equations is also possible, as program balances not only atoms, but also charge.īalanced chemical equation can be exported through the clipboard for use in other applications. The equation is already balanced - all you have to do is to read the result in the middle of the dialog window. Note that when you are entering formulas, molar masses of the reagents are calculated and displayed, saving you time and effort. Whilst sometimes it is necessary to use some fancy techniques to properly format formulas (as described in program help), in most cases they are not needed - just enter HClO4, P4O10, H3PO4 and Cl2O7. Simply enter formulas in the upper, input frame, into the formula edit fields. After this operation you are ready to enter reagents for equation balancing. At this moment there is not enough room for all reagents, so click once on the add reactant button and once on the add product button. Not much reagents, but relatively large number of atoms make balancing this chemical equation tedious, and it is so easy to make a mistake.Ĭreate a new reaction equation, by clicking on the New button on the tool bar. This is one of those chemical reactions that you see in your dreams the night before the exam. Changing a quiz from a past year is straightforward and I'm pretty much decided to purchase EBAS or perhaps the suite.īalance chemical equation: HClO 4 + P 4O 10 -> H 3PO 4 + Cl 2O 7 
I'm currently using EBAS when preparing quizzes and tests. Single user license price: €24.95 - approximately $25īy clicking Buy Now! you will continue to the FastSpring checkout page where payment will be taken, and your order fulfilled by FastSpring, our trusted reseller, and Merchant of Record.ĭownload 30-day free trial! 30-day money back guarantee! Equation balancer » example of balancing chemical equation 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
